Abdol A. Ameri, Weidenstetten
Das übergeordnete Ziel in der Behandlung der schubförmig remittierenden MS ist die Freiheit von klinisch und kernspintomographisch messbarer Krankheitsaktivität [5]. Alle Therapieentscheidungen – sowohl nach der Diagnosestellung als auch bei unzureichendem Ansprechen auf eine immunmodulatorische Behandlung – sollten unter Berücksichtigung der individuellen Patientenbedürfnisse, des Krankheitsverlaufs und des Nebenwirkungsprofils der verfügbaren Medikamente gemeinsam mit dem Patienten getroffen werden [5]. Da sich der Verlauf der schubförmig remittierenden MS in der Frühphase, das heißt bei EDSS(Expanded disability status scale)-Werten von<3, am besten beeinflussen lässt, wird ein möglichst frühzeitiger Beginn mit einer möglichst wirksamen Medikation empfohlen [6, 8].
Gut belegt ist die Wirksamkeit von Dimethylfumarat (DMF [Tecfidera®], 240 mg 2-mal täglich). Mittlerweile liegen Langzeitdaten über einen Beobachtungszeitraum von insgesamt sieben Jahren vor, die in den beiden zulassungsrelevanten, Placebo-kontrollierten Phase-III-Studien DEFINE und CONFIRM (zwei Jahre) und der offenen Anschlussstudie ENDORSE (fünf Jahre) gewonnen wurden [4]. Der klinische Nutzen war unabhängig davon, ob die Patienten neudiagnostiziert waren (definiert als Diagnosestellung innerhalb eines Jahres vor Einschluss in DEFINE/CONFIRM) oder zu Beginn der ENDORSE-Studie von Placebo auf DMF umgestellt wurden [4].
Langzeitwirksamkeit über sieben Jahre
In der Gruppe der neudiagnostizierten Patienten, die durchgehend über sieben Jahre mit DMF behandelt wurden (DMF/DMF), blieb die mittlere jährliche Schubrate über den gesamten Behandlungszeitraum stabil auf einem niedrigen Niveau von 0,13 Schüben/Jahr (Abb. 1) [4]. Nahezu zwei Drittel (61,8%) der Patienten blieben schubfrei. Der überwiegende Anteil der DMF/DMF-Patienten (85,4%) hatte auch nach sieben Jahren keine über 24 Wochen bestätigte Behinderungsprogression erlitten. Mehr als jeder Zweite (54,9%) blieb während der gesamten Beobachtungszeit ohne klinische Krankheitsaktivität [4]. Ein vergleichbarer klinischer Nutzen wurde auch bei den Patienten dokumentiert, die im Jahr 3 von Placebo auf DMF umgestellt wurden (PBO/DMF). Die kumulative Schubrate nahm signifikant um 61% ab (p<0,0001). Über die Hälfte der PBO/DMF-Patienten (55,3%) blieb schubfrei, 77,6% blieben frei von Behinderungsprogression und 48,2% ohne klinische Krankheitsaktivität [4].
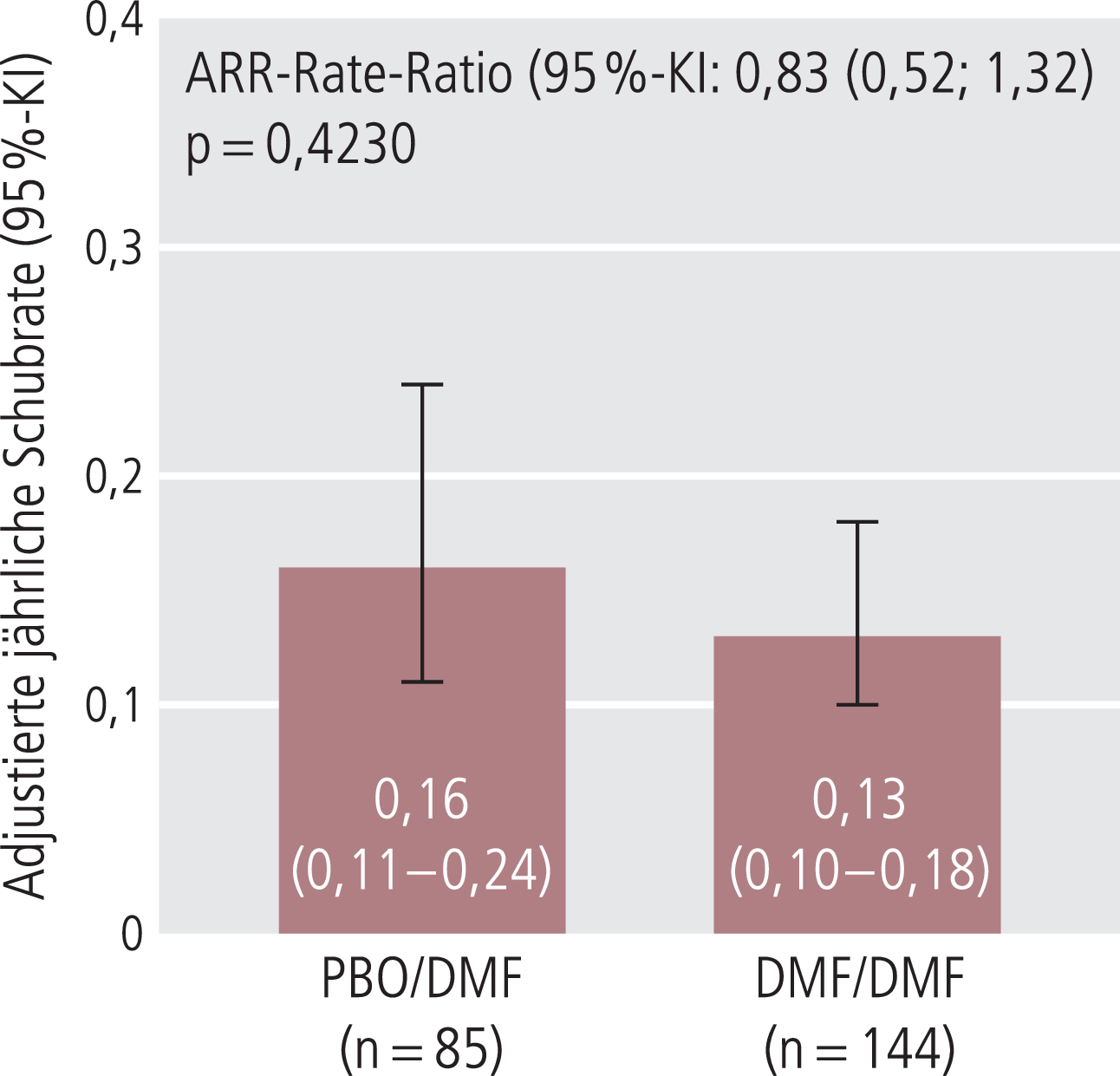
Abb. 1. Anhaltend niedrige mittlere jährliche Schubrate nach 7 Jahren unter kontinuierlicher DMF-Therapie (DMF/DMF) und nach Umstellung von Placebo auf DMF nach 2 Jahren (PBO/DMF) (mod. nach [4]). ARR: annualisierte Schubrate, KI: Konfidenzintervall
Registerdaten bestätigen die Wirksamkeit unter Praxisbedingungen
Zu ähnlichen Ergebnissen kam eine retrospektive Analyse der Behandlungsdaten von 8199 Patienten mit schubförmig remittierender MS, die in der klinischen Routineversorgung unterschiedliche krankheitsmodifizierende Therapien erhalten hatten [1]. Nach Adjustierung der Basischarakteristika war DMF mit einer signifikant niedrigeren jährlichen Schubrate in den 12 Monaten nach Therapiebeginn assoziiert als Interferon beta, Glatirameracetat und Teriflunomid. Die jährlichen Schubraten unter DMF waren vergleichbar mit denen unter Fingolimod [1]. Eine retrospektive Datenanalyse des globalen Registers MSBase bestätigt eine mit Fingolimod vergleichbare schubprophylaktische Wirksamkeit von DMF im klinischen Alltag [9]. Auch hier war das Schubrisiko unter DMF niedriger als unter Interferon beta, Glatirameracetat und Teriflunomid. In der DMF-Kohorte von MSBase waren nach zwei Jahren Therapie etwa 75% der Patienten schubfrei [9]. Zudem weisen Anwendungsdaten aus deutschen Arztpraxen darauf hin, dass eine Therapieumstellung auf DMF die jährliche Schubrate gegenüber einer Interferon-basierten Vortherapie um 72% und gegenüber einer Vortherapie mit Glatirameracetat um 78% reduzieren kann [7].
Praktikables Therapiemanagement mit DMF
Unter einer Behandlung mit DMF nehmen die Lymphozytenzahlen um etwa 30% ab und bleiben dann stabil [3]. Der Mittelwert bleibt bei der Mehrzahl der Patienten im Normbereich. Ein anhaltender Abfall der Zellzahlen auf Werte ≤0,5×109/l ist selten und wurde bei insgesamt 2,5% der Patienten beobachtet, die mehr als sechs Monate mit DMF behandelt wurden [3]. Um potenziell gefährdete Patienten rechtzeitig zu erkennen, wird empfohlen, vor Einleitung der DMF-Therapie und im weiteren Verlauf alle drei Monate ein Blutbild einschließlich Lymphozyten zu bestimmen [2].
Die Einnahme von DMF hat keinen relevanten Einfluss auf die pharmakokinetischen und pharmakodynamischen Eigenschaften oraler Kontrazeptiva mit den Wirkstoffen Norgestimat und Ethinylestradiol und umgekehrt [2, 10]. Der Wirkstoff hat eine Halbwertszeit von nur einer Stunde [2] und kann somit bei Kinderwunsch oder spätestens bei Vorliegen eines positiven Schwangerschaftstests kurzfristig abgesetzt werden.
Quelle
Prof. Dr. Ralf Gold, Bochum, Prof. Dr. Aiden Haghikia, Bochum, Pressekonferenz „Drei Jahre TECFIDERA® – Erweiterte Perspektive für MS-Patienten“, München, 14. Februar 2017; veranstaltet von Biogen.
Literatur
1. Boster A, et al. Annual relapse rates in multiple sclerosis patients treated with different disease-modify therapies – findings from a real world setting. ECTRIMS 2016; Abstract EP1481.
2. Fachinformation Tecfidera®, Stand: November 2016.
3. Fox RJ, et al. Absolute lymphocyte count and lymphocyte subset profiles during long-term treatment with delayed-release dimethyl fumarate in patients with relapsing-remitting multiple sclerosis. ECTRIMS 2016; Abstract P716.
4. Gold R, et al. Seven-year follow-up of the efficacy of delayed-release dimethyl fumarate in newly diagnosed patients with relapsing-remitting multiple sclerosis: integrated analysis of DEFINE, CONFIRM, and ENDORSE. ECTRIMS 2016; Abstract P631.
5. Gold R, et al. Therapieziele und Therapiemanagement bei schubförmig-remittierender multipler Sklerose. Nervenheilkunde 2015;34:915–23.
6. Leray E, et al. Evidence for a two-stage disability progression in multiple sclerosis. Brain 2010;133:1900–13.
7. Miclea A, et al. Safety and efficacy of dimethyl fumarate in multiple sclerosis: a multi-center observational study. J Neurol 2016;263:1626–32.
8. Scalfari A, et al. The natural history of multiple sclerosis: a geographically based study 10: relapses and long-term disability. Brain 2010;33:1914–29.
9. Spelman T, et al. Comparative analysis of MS outcomes in dimethyl fumarate-treated patients relative to propensity matched fingolimod, interferon, glatiramer acetate, or teriflunomide. ECTRIMS 2016; Abstract P1157.
10. Zhu B, et al. Delayed-release dimethyl fumarate does not adversely affect the pharmacokinetics of a commonly used oral contraceptive (norgestimate/ethinyl estradiol) in healthy women: Drug-drug interaction study results. AAN 2016; Abstract P2.097.
Psychopharmakotherapie 2017; 24(02)
